Cardiovascular disease progression extends beyond myocardial injury, as repeated injuries and vascular dysfunction lead to increased pressure and volume demands on the heart. This triggers a process known as remodeling, in which the heart adapts through structural and functional changes long before a clinical heart failure diagnosis. During this phase, termed cardiac stress and dysfunction, early vascular and myocardial injuries contribute to heightened hemodynamic burden and functional decline. A crucial biomarker in this context is N-terminal pro-B-type natriuretic peptide (NT-proBNP), which effectively reflects the biology of cardiac stress and can be measured at scale.1-5 This article emphasizes the importance of utilizing NT-proBNP within the cardiac biomarker continuum to investigate cardiac stress, remodeling, and the progression toward heart failure.
When the heart is exposed to sustained hemodynamic demand, myocardial wall stretch increases. Compensatory pathways are activated to preserve cardiac output and circulatory stability. While these responses are initially adaptive, persistent activation can contribute to chamber remodeling, stiffness, and declining cardiac efficiency.2,4
For research, this stage is important because cardiac stress biology often emerges before overt heart failure events. Stress markers, therefore, provide a way to study the transition from early vascular and injury biology toward functional decline, rather than focusing only on late clinical outcomes.2,3
NT-proBNP is released when heart muscle cells experience increased stretch and pressure. Within the cardiac biomarker continuum, it is best viewed as a signal of hemodynamic strain and workload, rather than direct cardiomyocyte injury.⁶
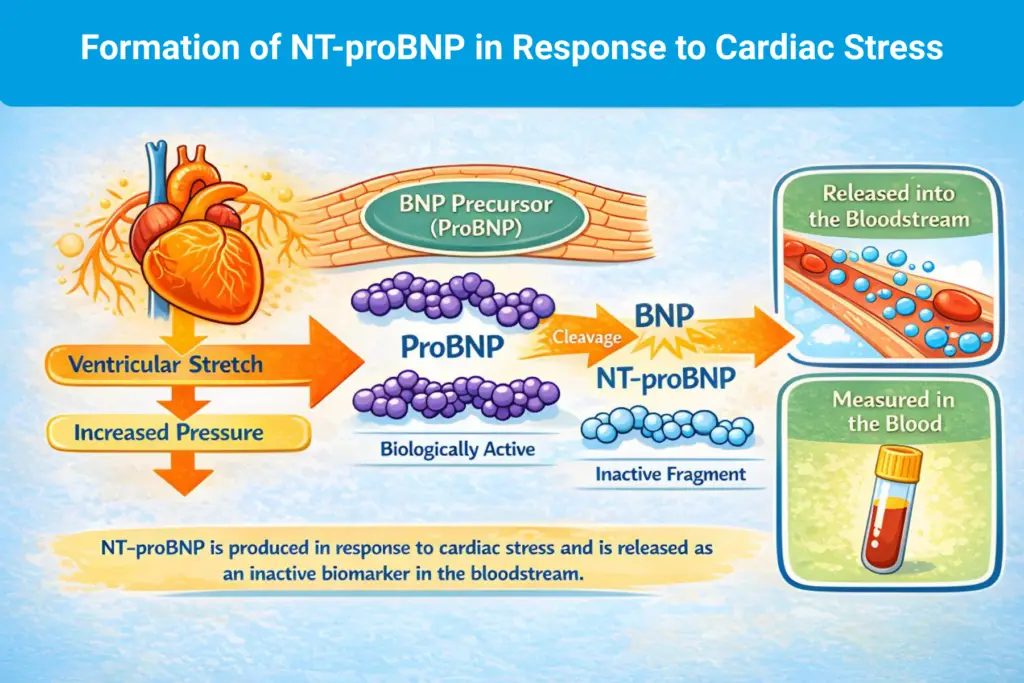
While high-sensitivity troponins reflect injury to heart muscle cells, NT-proBNP reflects stress and load biology. Together, they describe distinct dimensions of progression, enabling researchers to distinguish injury processes from compensatory stress responses.4,6
Heart failure can develop through multiple biological pathways, including ischemic injury, long-standing hypertension, metabolic disease, rhythm disturbances, and kidney disease. When studies rely only on broad heart failure outcomes, these diverse pathways can be grouped, reducing biological resolution within the study populations.1,2
NT-proBNP helps bring structure to this complexity by providing a consistent measure of cardiac stress. While it does not define underlying mechanisms on its own, it allows researchers to identify varying levels of stress burden within study populations and to study progression in a more biologically organized way.4,7
There are a variety of cardiovascular research studies that can be designed to utilize NT-proBNP when investigating cardiac stress, remodeling, and heart failure progression.
In longitudinal studies, repeated NT-proBNP measurements can help examine how cardiac stress evolves within individuals. Changes in NT-proBNP over time have been associated with structural changes and future outcomes. This supports its use for studying progression rather than single-time point risk.8
In intervention studies, NT-proBNP thresholds are frequently used to identify participants with clearer evidence of heart failure-related stress biology. This improves study efficiency and strengthens biological signal detection. NT-proBNP is also frequently measured during therapy to evaluate changes in stress burden over time, making it a practical tool for monitoring biological response in research settings.4,6,8
Earlier in this series, we highlighted high-sensitivity troponins as markers of myocardial injury. NT-proBNP complements these measures by capturing stress and workload. Together, injury and stress markers provide a more complete view of disease progression, linking subtle injury patterns to remodeling and functional decline.3,4
Across this series, several biomarkers have been positioned as tools to help study distinct stages of cardiovascular disease biology:
From a research design perspective, the cardiac biomarker continuum framework emphasizes that no single biomarker captures the full trajectory of cardiovascular disease. Studying a combination of markers that represent different biological processes can improve study design, increase biological resolution, and support longitudinal investigation of disease progression.2,3,4
By integrating vascular, injury, and stress markers, studies can move beyond event-based frameworks and toward models that reflect the full spectrum of disease progression, mechanisms, and transitions over time.2,5
No content on this site, regardless of date, should ever be used as a substitute for direct medical advice from your doctor or other qualified clinician.