Chronic kidney disease (CKD) is often described as a “silent” condition. In its early stages, structural damage can progress long before meaningful changes appear in conventional laboratory parameters. For researchers studying kidney injury and fibrosis, this creates a fundamental challenge: how do you detect and monitor progressive damage before significant functional decline is evident?
Emerging evidence suggests urinary Dickkopf-3 (DKK3) may help address this gap. By reflecting active tubular stress and fibrotic signaling, DKK3 provides insight into mechanisms driving CKD progression, making it a valuable biomarker in kidney research.
Dickkopf-3 (DKK3) is a glycoprotein that modulates the Wnt signaling pathway. The Wnt pathway plays a key role in embryonic development and is reactivated following organ injury. Experimental studies have shown that Wnt signaling is deeply involved in both acute and chronic kidney damage.¹
Temporary Wnt activation after injury can promote repair. However, sustained or dysregulated activation contributes to inflammation and fibrosis.²
Renal tubular cells under stress secrete DKK3, which is subsequently excreted in urine. Importantly, DKK3 is not detectable in the urine of individuals without kidney injury.⁷ This makes urinary DKK3 a direct reflection of ongoing tubular stress and profibrotic activity.
CKD is a growing worldwide health concern. The World Health Organization projects CKD to become the 5th most prevalent chronic disease by 2040.³ More than 800 million individuals worldwide are affected.⁴
Progressive CKD is characterized by tubulo-interstitial fibrosis, or progressive scarring of kidney tissue.⁶ Historically, fibrosis assessment has relied on biopsy and histopathology, which provide only a snapshot in time. Meanwhile, serum creatinine and estimated glomerular filtration rate (eGFR) reflect functional status at the time of testing but offer limited insight into future progression.
For researchers, this underscores the need for biomarkers that reflect active injury and ongoing fibrotic signaling, rather than static function alone.
DKK3 Reflects Active Tubular Stress
Tubular cells release DKK3 in response to acute or chronic injury. Its presence in urine signals ongoing kidney stress independent of the underlying cause.⁷
Because DKK3 is linked to profibrotic Wnt signaling, it provides mechanistic insight into the biological processes driving CKD progression.
Experimental and clinical models have demonstrated that urinary DKK3 levels correlate significantly with the extent of tubulo-interstitial fibrosis.²
Unlike biopsy, which captures a single time point, urinary DKK3 measurement enables longitudinal assessment of fibrotic activity in research settings.
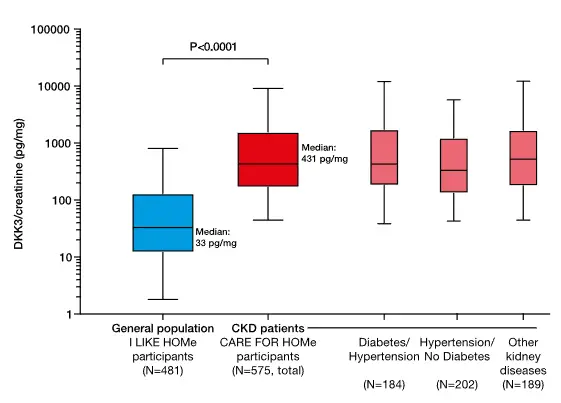
In a landmark study of individuals with CKD stages 2–4 followed over eight years, urinary DKK3 was an independent and significant indicator of short-term loss of kidney function.⁷
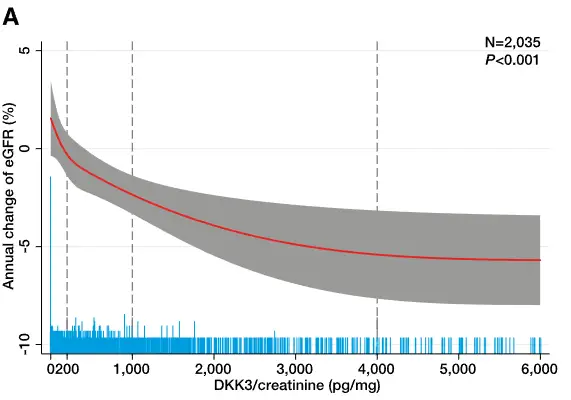
These findings have been demonstrated in adults⁷ and in children with CKD.⁹
When combined with eGFR, urinary DKK3 provides a more comprehensive picture of progression dynamics.
CKD progression is driven by fibrosis. Biomarkers that reflect this underlying biology are essential for advancing research into:
Because urinary DKK3 reflects active Wnt-mediated injury signaling, it aligns closely with the pathophysiology researchers aim to understand. It adds biological context beyond functional measurements alone.
The ReFiNE® DKK3 ELISA enables quantitative measurement of DKK3 in urine for laboratory research applications.
This assay supports investigators studying:
Discover more about the ReFiNE® DKK3 ELISA and how it can support your kidney research.
CKD often advances quietly, with fibrosis developing long before substantial functional decline is apparent. Unfortunately, traditional markers only reflect what has already happened.
Urinary DKK3, however, offers a different perspective: it reflects active tubular stress and profibrotic signaling, providing insight into the biological processes that drive progression. By integrating DKK3 measurement into CKD research, investigators gain a dynamic view of kidney injury that complements functional metrics like eGFR.
As research continues to unravel the complexities of kidney disease progression, biomarkers such as DKK3 may move the field from retrospective assessment toward forward-looking mechanistic understanding.
If you are exploring tools to enhance your CKD research, we’re here to help! Contact our Support Team to discover more about the capabilities of the ReFiNE® DKK3 ELISA today.
The ReFiNE® DKK3 ELISA is for research use only. Not for use in diagnostic procedures. For laboratory professional use only.
No content on this site, regardless of date, should ever be used as a substitute for direct medical advice from your doctor or other qualified clinician.