Cardiovascular disease (CVD) is not a single moment in time, but a biological progression that develops across years. Clinical events such as myocardial infarction (MI) and heart failure are late-stage outcomes along this pathway, rather than isolated occurrences. The “cardiovascular continuum” framework, articulated initially by Dzau and Braunwald and later expanded with clinical evidence, describes this evolution from early risk biology through vascular injury, ischemic events, remodeling, and end-stage disease.1-3 In this blog series, we aim to apply the cardiac biomarker continuum framework to cardiovascular research design across three biological stages: vascular inflammation and oxidative stress, myocardial injury, and cardiac stress and dysfunction.
Increasing evidence indicates that subclinical vascular and myocardial biology can precede symptomatic disease by years, creating an extended window during which disease processes are active but traditional event-focused testing may not capture them. Within this context, biomarkers provide practical research tools to investigate which biological pathways are engaged at different stages, enabling study designs aligned with disease progression rather than anchored solely to acute events.4
MI and heart failure are commonly used as endpoints in cardiovascular research; however, the mechanisms that lead to these outcomes often begin much earlier. The cardiac biomarker continuum model emphasizes that risk factors and early vascular injury can initiate a cascade of inflammation, oxidative processes, plaque development, and myocardial strain long before an acute presentation occurs. This long subclinical phase matters for research because it represents the stage where mechanistic insights, pathway monitoring, and early intervention hypotheses can be tested most directly.2,3
A cardiac biomarker continuum approach reflects the understanding that cardiovascular disease (CVD) progression involves multiple biological processes operating in parallel. Biomarkers serve as measurable readouts of these processes, including inflammatory activation, oxidative stress, cardiomyocyte injury, and hemodynamic stress. In this way, biomarkers function as pathway reporters rather than a single “answer” to a complex disease state.4, 5
No single biomarker captures the full trajectory of CVD. Instead, different biomarkers map to different biological domains (e.g., vascular inflammation vs. myocardial injury), and their interpretation is strengthened when they are positioned within the stage of disease biology being studied.
Because CVD is heterogeneous, investigations increasingly support combining biomarkers to capture complementary pathways and improve risk stratification and biological resolution. Reviews of multi-marker approaches describe additive diagnostic and prognostic value, particularly when biomarkers represent different mechanisms rather than redundant measures of the same pathway.5-7
For research design, the cardiac biomarker continuum is organized into three practical stages, each associated with distinct underlying biology:
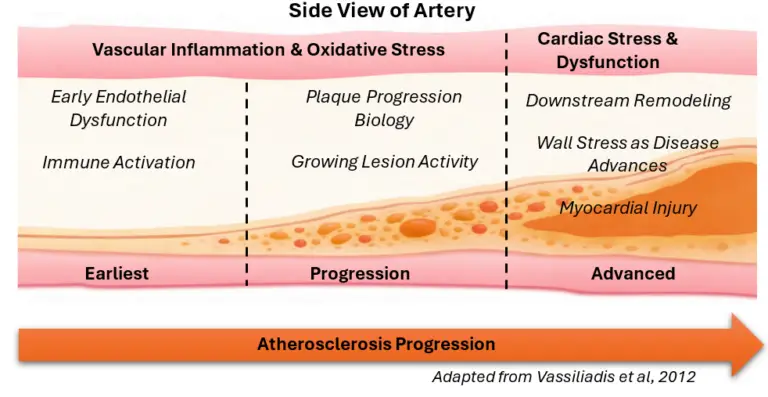
Earlier disease biology often produces minor changes in circulating biomarker concentrations, particularly in primary prevention cohorts or longitudinal monitoring studies. As a result, sensitive analytical methods can expand what is measurable, supporting detection of low-level myocardial injury and enabling more precise subgroup characterization in research populations. High-sensitivity troponin literature, for example, highlights how improved analytical sensitivity allows detection of myocardial injury at lower concentrations than earlier-generation assays.9-11
In parallel, cardiovascular research is increasingly shaped by large cohorts, repeated measures, and multi-marker strategies. High-throughput platforms support this shift by enabling routine monitoring and scalable workflows when studies require large sample sizes or higher-cadence sampling.
This blog series aims to apply the biomarker continuum framework to cardiovascular research design across three biological stages:
Together, these articles illustrate the continuum with biomarker examples, so researchers can understand not only whether disease is present, but which pathways are driving progression and how those pathways change over time.
No content on this site, regardless of date, should ever be used as a substitute for direct medical advice from your doctor or other qualified clinician.