Cardiovascular disease rarely begins with a dramatic clinical event. Long before myocardial infarction (MI) or heart failure, vascular biology shifts in subtle but meaningful ways: endothelial homeostasis is disrupted, inflammatory signaling increases, and oxidative pathways begin to reshape lipoproteins and the arterial wall. Within the cardiovascular biomarker continuum framework, this “early risk and vascular injury” stage is where researchers can most directly investigate how exposures, metabolic drivers, and immune activation initiate atherogenesis and sustain residual risk.1–3
In this phase, biomarkers are useful not because they help identify disease, but because they function as pathway readouts. They report on endothelial dysfunction, lipid oxidation biology, and inflammatory amplification in ways that traditional lipid panels often cannot. This article focuses on three complementary biomarkers to study vascular inflammation and oxidative stress: asymmetric dimethylarginine (ADMA), oxidized LDL (oxLDL), and myeloperoxidase (MPO).
The endothelium regulates vascular tone, barrier function, and thrombo-inflammatory balance. Under healthy conditions, nitric oxide (NO) signaling supports vasodilation and maintains an anti-inflammatory, antithrombotic vascular state. In early cardiometabolic dysfunction, however, NO bioavailability can decrease, and the endothelium shifts toward a more activated phenotype: more adhesive, more pro-inflammatory, and more permissive to plaque initiation.4
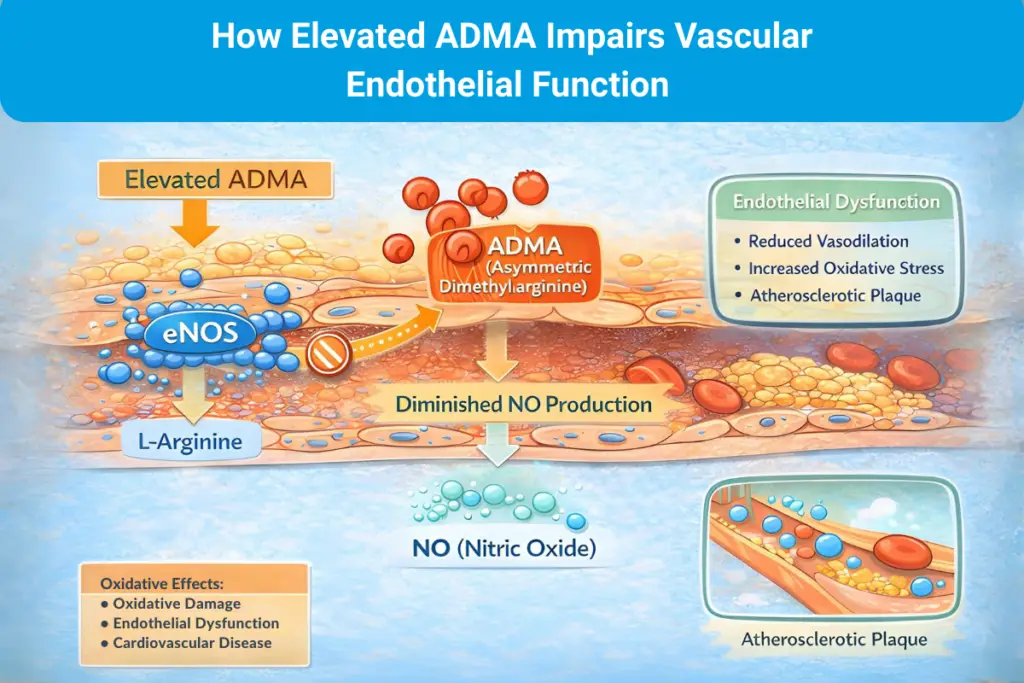
ADMA is frequently used in research as a mechanistic marker of this transition. As an endogenous inhibitor of nitric oxide synthase (NOS), elevated ADMA can reflect impaired NO production and, by extension, impaired endothelial function. In cohort studies and meta-analyses, higher ADMA has been associated with increased cardiovascular risk, supporting its use as a pathway-level biomarker in early risk biology rather than a late-stage damage marker.5
Traditional lipid panels (lipid profiles) measure blood lipid concentrations, including LDL-C, HDL-C, and triglycerides. Early atherogenesis, however, is not driven solely by LDL concentration. It is influenced by how LDL behaves within an inflammatory, oxidative arterial environment. Oxidized LDL (oxLDL) captures this mechanistic step: oxidative modification alters LDL particles in ways that promote immune activation, macrophage uptake, and foam cell formation, which are core features of plaque initiation and growth. 6
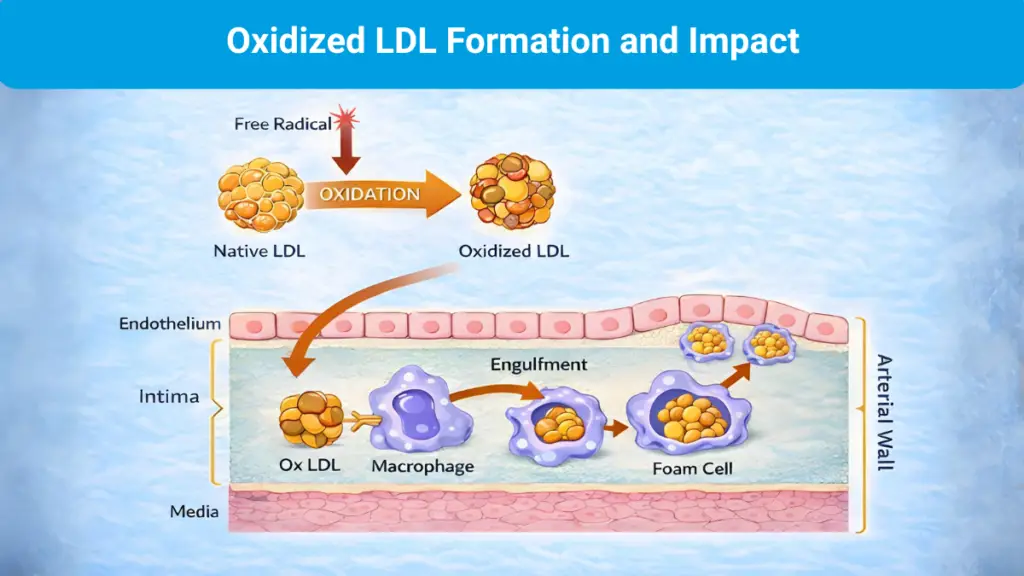
This distinction is important in research settings because LDL-C may appear “controlled” while oxidative and inflammatory pathways remain active. oxLDL can help clarify that biology by reframing the question from “how much LDL is present?” to “how is LDL being modified within an inflammatory, oxidative vascular environment?”6
If oxLDL is a mechanistic readout of lipid modification, MPO is a readout of inflammatory oxidative activity. MPO is a leukocyte-derived enzyme released by activated neutrophils and monocytes that generates reactive oxidants and promotes oxidative modification of proteins and lipoproteins within atherosclerotic tissue. It is widely described as both a marker of inflammatory activation and an active contributor to oxidative pathways linked to plaque progression and instability.7
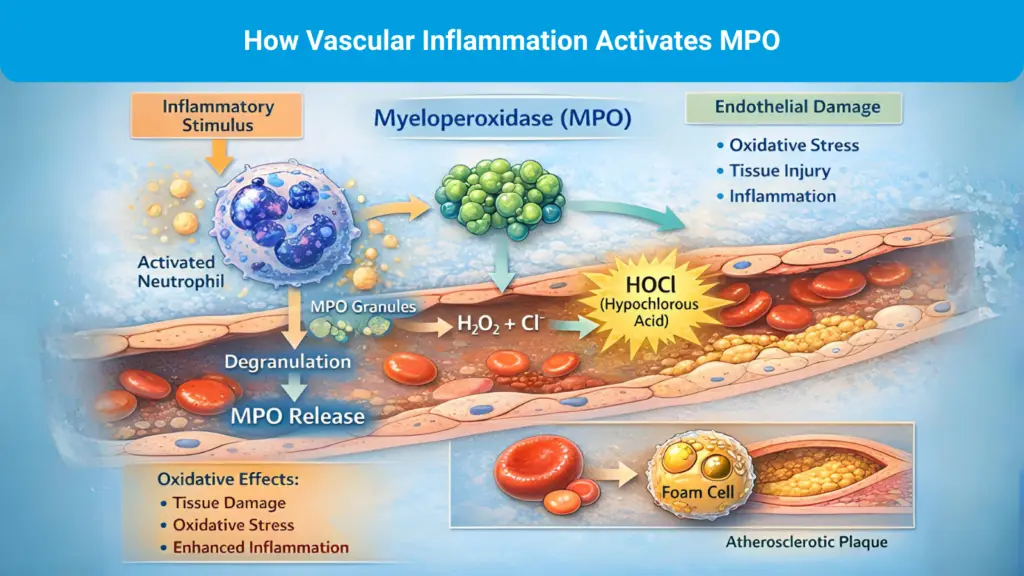
Clinically anchored studies also support MPO’s relevance to cardiovascular risk. In subjects presenting with chest pain, MPO has been shown to predict adverse outcomes even when troponin is negative at baseline, suggesting MPO reflects upstream vascular inflammatory activity rather than cardiomyocyte necrosis.8
Lipid panels remain essential in clinical care, but they have limitations as research tools when the goal is to understand mechanisms. They primarily quantify concentration, not pathway activity. oxLDL and MPO help expose oxidative and inflammatory biology that can persist even when LDL-C appears similar across individuals. ADMA adds a complementary dimension by reporting on endothelial NO-pathway dysfunction, an upstream shift that can precede overt plaque or symptomatic disease.4–7
Taken together, these biomarkers support a more mechanistic approach to early-stage cardiovascular research: not simply who is at risk, but which pathways are active, how early they appear, and how they evolve.
The early vascular stage is the start of the biomarker continuum and sets the conditions for what comes next. Endothelial dysfunction, lipid oxidation, and inflammatory amplification shape the arterial environment, increasing the likelihood of ischemia and downstream cardiomyocyte injury. ADMA, oxLDL, and MPO are essential biomarkers for studying vascular inflammation and oxidative stress. By investigating these markers rather than traditional lipid panels, researchers can study pathway activity mechanistically, enabling a more complete understanding of cardiovascular disease progression.
In our next article, we focus on myocardial injury markers, especially high-sensitivity troponins, used not only to detect acute MI but also to study subclinical injury patterns and link injury signals to outcomes in longitudinal cohorts. We also shift from vascular biology to cardiomyocyte biology and discuss how researchers can interpret injury biomarkers across acute and subclinical contexts.
No content on this site, regardless of date, should ever be used as a substitute for direct medical advice from your doctor or other qualified clinician.