The microbiome–gut–brain axis plays a central role in irritable bowel syndrome (IBS) by linking microbial activity to neurological and immune signaling pathways. Measuring stool-based neurotransmitters, such as serotonin, GABA, histamine, and tryptophan, provides functional insights into this bidirectional communication. These biomarkers enable researchers to better understand IBS mechanisms, symptom variability, and potential therapeutic targets1,2.
Irritable bowel syndrome is a complex, multifactorial disorder characterized by abdominal pain and altered bowel habits. It affects an estimated 10–15% of adults in the United States3. This prevalence makes IBS a key focus for researchers studying gastrointestinal physiology and systemic signaling pathways.
The microbiome–gut–brain axis describes the continuous, bidirectional communication between the gut and the brain.
The central nervous system (CNS) communicates with the intestine through the autonomic nervous system (ANS), including afferent and efferent nerve pathways1. These signals regulate intestinal motility, permeability, immune activity, and mucus secretion, all of which influence microbiome composition.
In parallel, the gut microbiome produces signaling molecules that act locally and systemically. These include neurotransmitters and metabolites that influence enteric nervous system activity, immune responses, and central nervous system signaling1,2.
Disruptions in this communication network can alter both gastrointestinal function and neurological processing, contributing to the development of IBS symptoms.
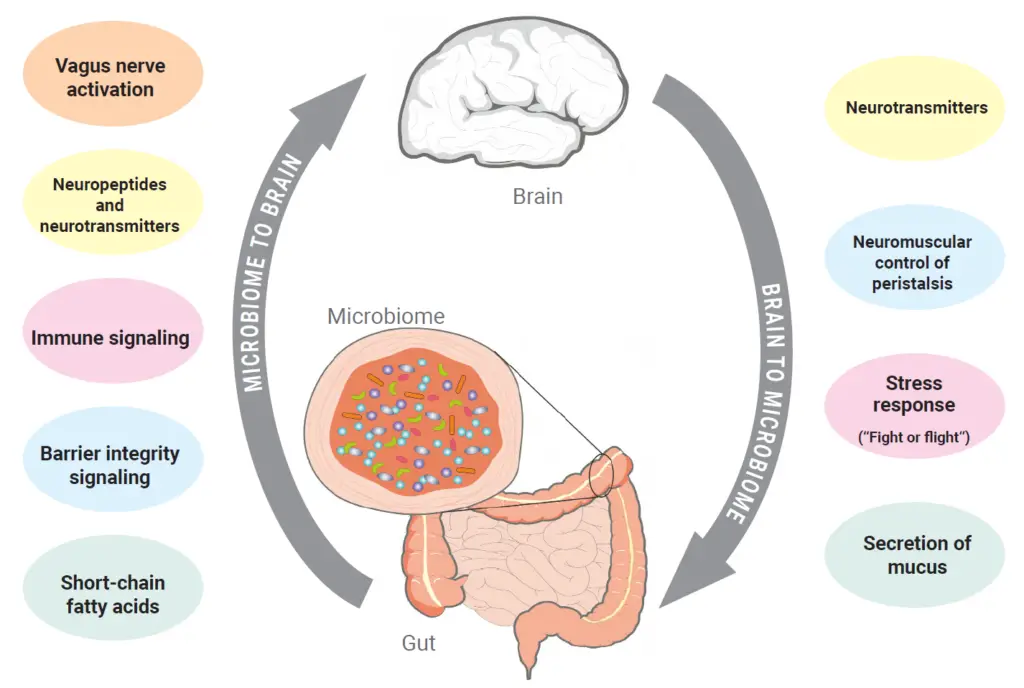
The gut microbiota produces bioactive compounds that function as neurotransmitters, tissue hormones, and growth factors.
Key molecules include GABA, serotonin, histamine, and tryptophan, which regulate:
These molecules integrate microbial metabolism with host signaling pathways, making them highly relevant to understanding IBS subtypes and symptom variability1.
Histamine is a tissue hormone involved in immune regulation and inflammatory signaling.
It is produced by mast cells, enterochromaffin cells, and certain gut bacteria. Histamine regulates cytokine production, including IL-6 and TNF-α, and supports immune cell recruitment4.
An imbalance between histamine production and degradation by enzymes such as diamine oxidase (DAO) and histamine-N-methyltransferase (HNMT) can lead to accumulation. Elevated histamine levels have been associated with symptoms such as diarrhea, nausea, headaches, and tachycardia5.
In IBS, histamine may reflect immune-driven symptom pathways and altered mucosal responses.
Serotonin is a key regulator of gut motility and sensory signaling.
It controls intestinal peristalsis through the 5-HT3 receptor6. Low serotonin levels are associated with constipation-predominant IBS (IBS-C), while elevated levels are associated with diarrhea-predominant IBS (IBS-D)7.
Excessive serotonin signaling, combined with receptor overexpression, may contribute to abdominal pain and hypersensitivity8.
Because most serotonin is produced in the gut, it is a critical biomarker for microbiome-driven functional changes.
Tryptophan is an essential amino acid that serves as a precursor to serotonin and plays a key role in intestinal barrier function.
It activates mTOR signaling pathways, leading to increased production of barrier proteins, defensins, and secretory IgA9. Tryptophan is also metabolized into indole derivatives that exert anti-inflammatory effects10.
Altered tryptophan metabolism has been linked to immune imbalance and mucosal dysfunction in IBS.
Gamma-aminobutyric acid (GABA) is an inhibitory neurotransmitter involved in regulating visceral pain.
It is produced by both host tissues and gut microbes. GABA reduces neuronal excitability and modulates pain signaling pathways11.
Lower GABA levels have been associated with increased pain sensitivity in IBS patients12,13.

Stool-based measurement of microbiome-derived metabolites provides a non-invasive and functionally relevant way to assess gut–brain axis activity.
Analyzing neurotransmitters and tissue hormones in stool enables researchers to:
This functional approach provides deeper insight than microbiome composition alone2.
Because neurotransmitters such as serotonin, GABA, histamine, and tryptophan reflect microbiome activity, quantifying them in stool provides a functional readout of gut–brain signaling.
Research suggests that identifying the neurotransmitters and tissue hormones produced by bacteria in stool can provide essential insights into disturbances in communication between the brain and the gut microbiome, such as in irritable bowel syndrome.
Immundiagnostik, Inc. offers a panel of ELISAs designed for research use to measure these key biomarkers:
These ELISAs enable reproducible, quantitative measurement of microbiome-derived signaling molecules, allowing researchers to generate actionable insights into IBS pathophysiology.
The microbiome–gut–brain axis is a central mechanism in IBS, linking microbial metabolism to neurological and immune signaling pathways.
Measuring stool-based neurotransmitters such as serotonin, GABA, histamine, and tryptophan provides a direct and functional view of this communication. ELISA-based biomarker analysis enables researchers to move beyond descriptive microbiome studies toward mechanistic insights.
By integrating these biomarkers into IBS research workflows, investigators can better characterize disease subtypes, understand symptom drivers, and explore new therapeutic strategies.
All Products are for Research Use Only. Not for use in diagnostic procedures. For laboratory professional use only.
No content on this site, regardless of date, should ever be used as a substitute for direct medical advice from your doctor or other qualified clinician.